Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Formic Acid Feed Grade
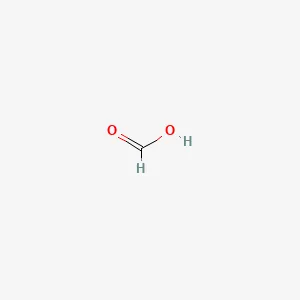
Formic Acid Feed Grade Structure
| Chemical Name | : Formic Acid |
| Common Names | : Methanoic Acid, Hydrogen Carboxylic Acid, Aminic Acid |
| CAS Number | : 64-18-6 |
| Molecular Formula | : HCOOH (CH₂O₂) |
| Molecular Weight | : 46.03 g/mol |
| HS Code | : 2915.11 |
| Grade | : Feed Grade |
Formic Acid Feed Grade is the simplest carboxylic acid, naturally occurring in the venom of ants and in various fruits. In the animal feed industry, it is widely used as an antimicrobial preservative and acidulant due to its potent activity against a broad spectrum of bacteria, molds, and yeasts.
When added to animal feed or drinking water, formic acid lowers the pH of the gastrointestinal tract, creating an unfavorable environment for pathogenic microorganisms such as Salmonella, E. coli, and Campylobacter. This improves feed hygiene, extends shelf life, and promotes better nutrient absorption by the animal.
Formic acid feed grade is a clear, colorless liquid with a pungent odor, available in concentrations typically ranging from 85% to 99%. It is used either in pure form or in blended acid formulations tailored for specific species and production stages.
| Appearance | : Clear, colorless liquid |
| Odor | : Pungent, characteristic |
| Boiling Point | : 100.8 °C |
| Melting Point | : 8.4 °C |
| Density | : 1.22 g/cm³ (at 20 °C) |
| Solubility in Water | : Miscible in all proportions |
| pH (1% solution) | : ~2.3 |
| pKa | : 3.75 |
| Purity (Feed Grade) | : ≥ 85% |
| Flash Point | : 69 °C (closed cup) |
Feed Preservation: Prevents mold and bacterial growth in silage, compound feed, and raw materials, extending shelf life and maintaining nutritional quality.
Drinking Water Acidification: Added to poultry and swine drinking water to reduce microbial load and improve gut health.
Salmonella Control: Effective in reducing Salmonella contamination in feed, supporting food safety from farm to fork.
Digestibility Enhancement: Lowers stomach pH in young animals (piglets, chicks), aiding protein digestion and improving feed conversion ratio.
Silage Production: Used as a silage additive to rapidly lower pH and preserve fermentation quality.
Formic acid is produced industrially via the methyl formate process: carbon monoxide reacts with methanol under pressure to form methyl formate, which is then hydrolyzed to yield formic acid and methanol. The methanol is recycled. An alternative route involves the oxidation of formaldehyde. Feed-grade product undergoes additional purification and quality testing to meet regulatory standards.
Formic acid is a corrosive substance and must be handled with appropriate care.
Flammable liquid (Cat. 4); Acute toxicity oral (Cat. 4); Skin corrosion (Cat. 1A); Serious eye damage (Cat. 1).
Danger
H227 – Combustible liquid H302 – Harmful if swallowed H314 – Causes severe skin burns and eye damage H331 – Toxic if inhaled
Vapors are irritating to the respiratory tract; high concentrations may cause pulmonary edema.
Causes corrosive burns; prolonged contact may result in severe tissue damage.
Causes serious, potentially irreversible damage to the eyes.
Immediately move to fresh air. If breathing is difficult, administer oxygen. Seek emergency medical attention immediately.
Remove contaminated clothing immediately. Flush skin with large amounts of water for at least 20 minutes. Do not neutralize chemically. Seek urgent medical attention for burns.
Immediately flush eyes with large quantities of water for at least 20 minutes, keeping eyelids open. Seek immediate medical attention; risk of permanent eye damage.
Do not induce vomiting. Rinse mouth and drink 1–2 glasses of water. Seek immediate medical attention.
Acid-resistant gloves (e.g., neoprene or butyl rubber)
Chemical splash goggles and face shield
Acid-resistant apron and full body protection
Respiratory protection (half-mask respirator with organic vapor/acid gas cartridge) in poorly ventilated areas
Handle only in well-ventilated areas or under local exhaust ventilation to control vapor exposure.
Avoid contact with skin, eyes, and clothing. Use full PPE at all times during handling.
Keep away from heat sources, sparks, and open flames due to combustibility.
Use acid-resistant equipment (polyethylene, polypropylene, or stainless steel); avoid aluminum, zinc, and copper fittings.
Do not mix with strong oxidizing agents, strong bases, or reactive metals.
Store in a cool, dry, well-ventilated location, away from heat sources and direct sunlight.
Keep in tightly closed, corrosion-resistant containers (polyethylene, stainless steel).
Separate from incompatible materials: strong oxidizers, bases, and reducing agents.
Recommended storage temperature: 10 – 30 °C.
Shelf life: 24 months from date of manufacture under recommended storage conditions.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product